Mycometer® surface Fungi
For Quantification of Fungi on Surfaces
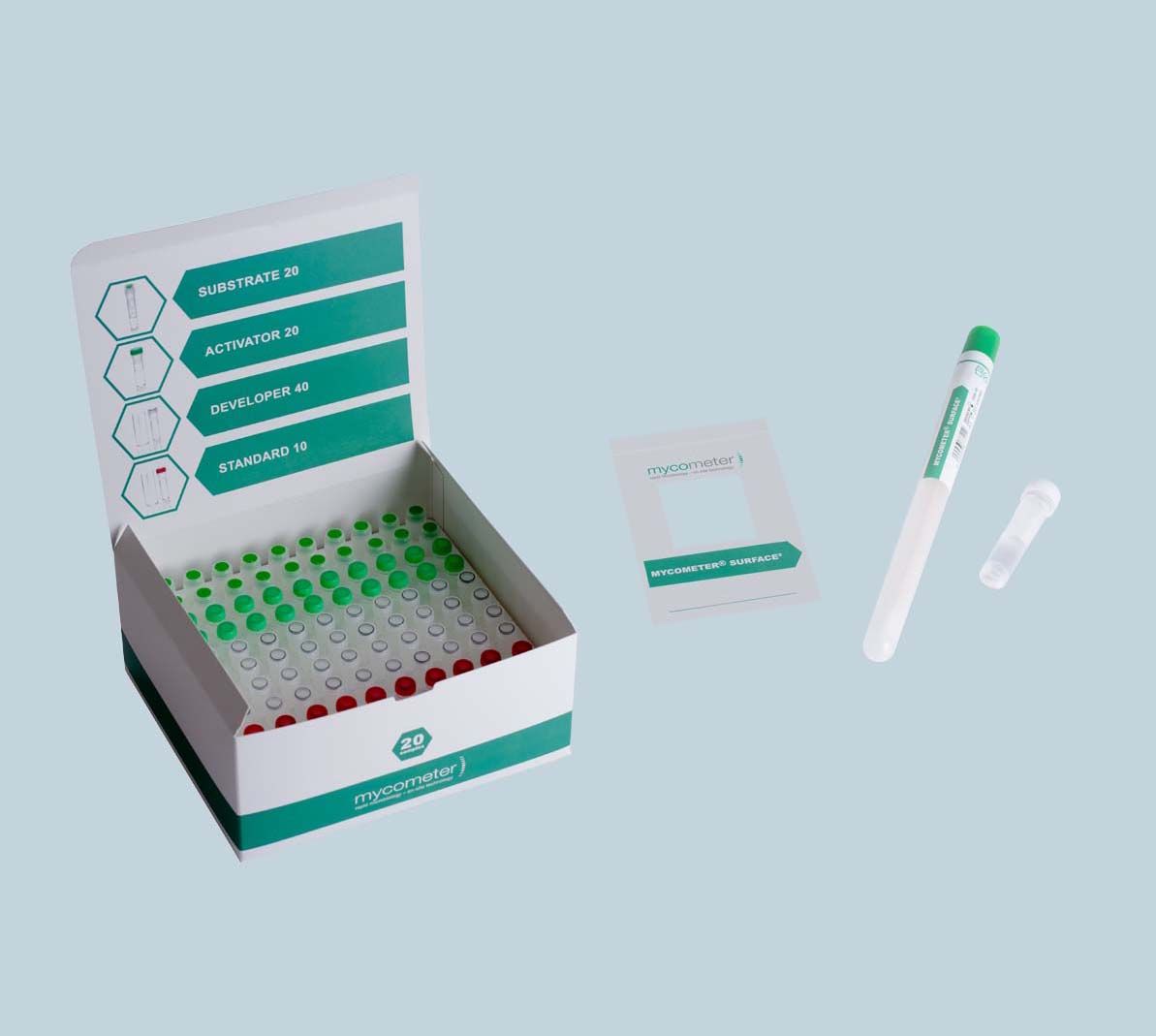
Test Assays
The tests assays are packed in a box of 20 samples. A box contains all accessories for sampling and analysing 20 samples.
20 Sampling units
20 Substrates
20 Activators
40 Developers
10 Fluorescent standards
2 Filtration units
40 Pipette tips (100µl)
Product Information
Quantification of fungi on surfaces in less than an hour
The methodology has been developed in collaboration with mould remediation consultants and contractors and the Danish Building Research Institute, and has been proving its value for more than a decade.
Results categories have been established empirically, and provide criteria for clean, dirty and contaminated.
Clean is the defined as the background level meaning the level found on visual clean surfaces in well maintained building with no mould and moist problems.
Contaminated is defined as samples containing mould in higher concentration than what can be found on dusty and dirty surfaces in well maintained building with no mould or moisture problems.
The interpretation criteria are in accordance with the most influential international guidelines such as those from WHO, New York City Department of Health and US-EPA.
All mould growth, independent of species can affect health and should be removed. Protocols have been developed for measurement of mould contamination inside porous materials. Insulation materials, materials containing cement (e.g. concrete, plaster, mortar) and other porous materials found in buildings can contain mould growth deep inside.
The Mycometer technology has been tested daily for more than 15 years by consultants and contractors in tens of thousands of mould remediation projects.
Several independent scientific papers have documented the principle behind the Mycometer technology, and in 2011 the technology was verified by the US-EPA (US-Environmental Protection Agency).
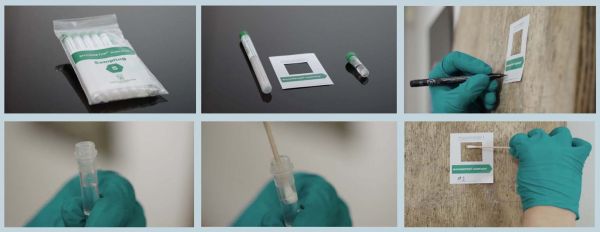
- Break the sterile seal on the swab container by twisting the cap.
- The cotton swab is then wetted in the sterile saline contained in the green capped tube.
- Apply the self-adhesive template to the surface to be sampled.
- Firmly grip the wooden swab handle, keeping your fingers at least one inch from the cotton swab.
- Thoroughly wash the surface by rubbing the area inside the template. The swab should be rotated in order to use as much of the cotton as possible.
- Keep a low angel between the swab and surface.
- Replace the swab into the container until analysis.
The samples can be stored up to one week before analysis. The sterile saline contains a biostatic agent that prevents any growth in the sample during storage. However, it is recommended that samples be analyzed as soon as possible after sampling.
Porous materials
Insulation materials: Most insulation materials although primarily inorganic, contain organic compounds which can support mould growth (e.g. binders).
Dust and debris including spores from outdoors may supplement the level of organic materials, naturaly accumulating over time. Therefore, when in the presence of moisture such as humidity or wetting, insulation can sometimes contain actual mould growth.
In our investigation we found that visual assessment was inadequate for evaluating the insulation material. Sometimes insulation material that looks clean may contain high levels of mould growth while dirty looking materials do not.
Cementitious materials: New cementitous materials are naturally protected from mould growth due to the high alkalinity (pH 11.5-12.5). However, due to carbonization, where the Ca(OH)2 in the cement reacts with the CO2 in the air producing CaCO3, pH lowers over time with an ending point of around pH 7-8. High levels of mould can be found even though the materials looks solid and has no visual discoloration indicating water damage. Investigation of porous materials:
In porous materials, the vast majority of the mould growth can often be found inside the material rather than on the surface.
Analyzing bulk samples with the Mycometer test allows measurement of this hidden mould source, something no other method can provide. The bulk samples are weighed and then simply put into the enzyme substrate solution instead of a swab.
The time of analysis is the same as with swab samples. The results are given as fluorescence counts per mg material.
Examples of relevant materials for bulk sampling are:
- Rigid fibrous glass duct liner
- Insulation material e.g. mineral wool and glass wool
Ceiling Tiles Plaster – Mortar – Concrete Protocols are available for some materials, e.g. insulation materials and in materials containing cement (mortar/plaster/concrete).
Protocols are available to download from our password-protected customer portal.
The Mycometer products are based on a well-documented technology. Independent, as well as scientists from Mycometer have published and are currently publishing papers on the technology in peer reviewed well reputed international journals and at scientific conferences. The technology has gone through the ETV (Environmental Technology Verification) program United States Environmental Agency (US-EPA´s). However, the best documentation is the fact that our customers keep coming back for more products and sales have been increasing every year since we started.
Publications
- Airborne fungal cell fragments in homes in relation to total fungal biomass. Adhikan A, Reponen T. Rylander R. Indoor Air 2012; in print.
- Nocturnal asthma and domestic exposure to fungi. Terčelj M, Salobir B, Narancsik Z, Kriznar K, Grzetic-Romcevic T, Matos T, Rylander R. Indoor + Built Env. 2012; in print.
- Enzyme measurements for risk evaluations in sewage treatment plants. Rylander R, Calo A. Open J Publ Health, Jun 2012.
- Beyond LEED, Pre and Post Occupancy Evaluations for New Buildings. P Buckmaster. Synergist, May 2011.
- Assessment of the Bacterial Contamination and Remediation Efficacy after Flooding Using Fluorometric Detection. M. Reeslev, JC Nielsen, L Rogers. Accepted for publication ASTM Journal, 2011.
- Aggressive Sampling, Improving the Predictive Value of Air Sampling for Fungal Aerosols. M. Reeslev, M. Miller, JC Nielsen, L Rogers. Proceedings of Indoor Air Conference, ISIAQC. June 2011, Austin Texas.
- Airborne enzyme in homes of patients with sarcoidosis. Terčelj M, Salobir B, Rylander R. Env Health 2011; 10; 8-13.
- Airborne enzyme measurements for the identification of mouldy buildings. Rylander R, Reeslev M, Hulander T. . . J Environ Monit, 2010; 12:2161-2164.
- Fluorometric detection and estimation of fungal biomass on cultural heritage materials. Journal of Microbiological Methods 80 (2010) 178–182, R Mitchell, et al (Harvard) 2010.
- Airborne enzyme measurements to detect indoor mould exposure. Journal of Environmental Monitoring, V.12, p.2161–2164. R. Rylander, et al. 2010.
- Successful Mold Growth Remediation in HVAC Systems. P Buckmaster. Occupational Health and Safety, January 2008.
- Quantifying Mold Biomass on Gypsum Board: Comparison of Ergosterol and Beta-N-Acetylhexosaminidase as Mold Biomass Parameters. Applied and Environmental Microbiology. Vol. 69, No.7, p. 3996-3998. M.Reeslev, M.Miller, KF Nielsen. 2003.
- Analytical Instrument Performance Criteria: Application of a Fluorometric Method for the Detection of Mold in Indoor Environments. Applied Occupational and Environmental Hygiene. Vol. 18, No.7, p. 499-503. D Krause, YY Hamad, L Ball. 2003.
- Application of a Fluorometric Method for the Detection of Mold in Indoor Environments. (2003), D. Krause. Applied Occupational and Environmental Hygiene Volume 18(7): 1–5.
- Nagase Activity in Airborne Biomass Dust and Relationship between Nagase Concentrations and Fungal Spores. Aerobiologia Vol. 19, 97 – 105. A.M., Madsen. 2003.
- The Mycometer™-Test: A New Rapid Method for Detection and Quantification of Mold in Buildings. Proceedings of Healthy Buildings 2000, Vol. 1, p.589-590. M.Reeslev and M. Miller. 2000.
- Fluorogenic Substrates to Measure Fungal Presence and Activity in Soil. Appl. Environ. Microbiol. 64:613-617. M. Miller, A. Palojärvi, A. Rangger, M. Reeslev, A. Kjøller. 1998.
Interpretation criteria are available for differentiating surfaces containing mould growth from those containing deposited spores as on a dusty surface, and for defining clean as the success criteria for the cleaning of surfaces.
Interpretation criteria is also available for measuring inside porous materials such as insulation materials (glass wool, mineral wool, ceiling tiles and other) and in cement containing materials (mortar, some types of concrete). The criteria allow differentiation of contaminated materials (containing mould growth) from non-contaminated materials.
Data has been collected from hundreds of samples to establish the interpretation criteria. These categories of surface contamination were developed as a result of testing performed in collaboration with the Danish Building Research Institute and the Technical University of Denmark.
Criteria for Mycometer surface Fungi
Samples collected on a 9 cm2 area and analysed according to the Mycometer surface Fungi protocol.

- Post remediation verification (PRV)
- Documenting the efficacy of the cleaning of surfaces for mould growth
- Documentation of mould growth
- Documentation that discoloration is NOT mould growth
- Delineation of mould growth where it is non-visible. How much should be cleaned?
- Documenting the cleaning of HVAC systems
Universities and government agencies
The Danish Technical University – DTU (Denmark)
The University of Eastern Finland (Finland)
US Army CHPPM-South, (Korea)
US Army CHPPM South ( Japan)
The Technological Institute of Denmark (Denmark)
US Army- Ft Eustis, Ft Drum, Ft Meade ++ (US)
US Navy – Camp Pendleton, Annapolis ++ (US)
•Instituto de patologia del libro (Italy)
Danish Army Building Services (Denmark)
The Finnish Institute of Occupational Health (Finland)
Defense Intelligence Agency (US)
The Royal Danish Academy of Fine Arts. The School of Conservation (Denmark)
Hochschule für angewandte wissenshaft und Kunst (Germany)
Corps of Engineers (US)
Institut National de Researche et de Securite – INRS (France)
National Security Agency – NSA (US)
CDC/NIOSH (US)
National Institute for Health and Welfare (Finland)
National Institutes of Health -NIH (US)
Institut de recherche Robert-Sauvé en santé et en Sécurité du travail –IRSST (Canada)
Consultants and contractors
EES Group (US)
CETEC (Australia)
National Flood School (UK)
Golder Associates, Inc.(US)
COWI (Denmark)
Regas Group (US)
Goritas building inspection (Denmark)
Building Health Sciences, Inc. (US)
Action Dry Emergency Services (UK)
Industry- and Consultant Services (Norway)
Inuplan (Greenland)
AMiP – Industrial Engineering GmbH (Austria)
Air Purification Specialists (US)
BioFact (Sweden)
Dominion Environmental (US)
ISS (Denmark & Finland)
Docusave (Switzerland)
R&T (Greenland)
Inniluf Tænastan (Faroe islands)
Environ, Inc. (US)
BBJ Environmental Solutions (US)
LBF (Faroe Islands)
Indoor Air Sciences, Inc. (US)
Robson Environmental ( Australia)
ROSE Environmental, LTD ( Trinidad)
ArQi:Mate ( Singapore)
PT Freeport Moran Mining (Indonesia)
Engineering Systems, Inc. (FL, TX) (US)
SSG (Denmark)
National Analytical Labs (US)
Accredited laboratories
ita Laboratorio Oy (Finland)
ISS Proko Oy (Finland)
MetropoliLab Oy (Finland)
FAQ
Yes. Time is MONEY. When there is no need to wait for laboratory results, remediation work can be started quickly, allowing for quick revitalization and rapid reuse of land / areas. In addition, the Mycometer Surface Test can measure the efficiency of certain methods used to clean contaminated materials. The use of the Mycometer Surface Test allows remediators and consultants to quickly and empirically determine whether or not materials can be cleaned.
While we cannot predict the outcome of litigation in court, we can report the latest outcome of proceedings in Texas and Florida courts. Expert opinions based on Mycometer samples were accepted in three Florida courts and six Texas courts. The widespread acceptance of Mycometer test results by U.S. federal agencies such as the Department of Defense or state institutions (e.g., the Florida and Texas Departments of Health) illustrates the acceptance of the method by scientific experts. The acceptance of Mycometer depends on expert opinions based on the data provided. Overinterpretation endangers the acceptance of any test method.
Cultivation methods such as agar plates, RODAC plates or dipslides can be used for the quantification of fungal spores, which are usually (with few exceptions) unicellular. One cell = one colony. However, mold consists of multicellular hyphae, so that the principle “one cell = one colony” cannot be applied. Since spores typically consist of only 0-5% of the total fungal biomass present in mold, cultivation methods are not well suited to quantify fungal biomass (mold). The Mycometer test quantifies mold by measuring the enzyme activity present in both spores and hyphae in approximately the same amount per unit of biomass.
If you take “fair” as confirmation or evidence that the contractor has fulfilled the task of returning materials to an uncontaminated state, then Mycometer is absolutely fair. However, it is fair to say that Mycometer is a very accurate, unadulterated test that is more difficult for some inefficient contractors to handle than previously applied air sampling or visual inspection.
Adenosine triphosphate (ATP)-based tests are often very inexpensive, fast and easy to use, but they are much less reliable than Mycometer-based methods. Overall, Mycometer differs greatly from ATP-based methods, mainly for the following reasons:
1. the ATP molecule is present in all living organisms, which is not the case with the enzyme ((N-acetylhexosaminidase (NAHA)) measured in the Mycometer test.
2. the thick-walled fungal cells must be opened to measure ATP, while no extraction is necessary to measure NAHA. The ATP molecule is not fungus-specific, it is present in all living organisms, in cells as well as in exudates and secretions. Enzyme activity (NAHA, which is detected by the Mycometer method, even if it is not purely fungus-specific, is a much more specific evidence for the presence of fungal infection.
An example: Wipe a surface with a finger. This will leave large amounts of ATP on the surface, while no NAHA can be detected.
It is often argued that ATP can at least be used to test whether surfaces have been successfully cleaned, although it is not fungus-specific: if there is no ATP on the surface, there is no more mold on the surface. On the surface this sounds logical and correct – but it is not. To measure ATP from fungal cells, the cell walls must be opened and the ATP must be extracted. In most ATP studies, no chemicals are added to open the cell walls. And even if this were done, it would take a relatively long time to open the cells. As a result, the available ATP tests only measure a small amount of ATP in the fungus and thus lead to false, negative results. In other words, mold may be present without being detected by ATP tests.
In addition, the Mycometer test has been approved by the U.S. Environmental Protection Agency (EPA), an independent agency that has found the method to be highly reproducible. To the best of our knowledge, there are no ATP methods or procedures recognized by the US EPA.
Mycometer results perform well in relation to adhesive tape. This is especially true when the microscopy is performed by only one person. Both methods aim to measure fungal biomass, both hyphae and spores. Due to the subjective nature of microscopy, which is subject to personal interpretation, the two methods do not always correlate well. A more detailed discussion of this correlation can be found in the paper “”Measuring the efficacy of Mold Remediation on Contaminated Ductwork” by J.D. Krause and Y.Y. Hamad.
Anyone can learn to perform the analysis. However, all those who use the Mycometer procedure must undergo training. A basic course is available on the Internet or on USB stick. This course contains instructions on all the basic steps of sampling and analysis and ends with a final exam. On-site training is more comprehensive. It is an interactive course with a high practical component. On-site training is available for Mycometer-Surface, Mycometer-Air and for bulk solids (as well as for Bactiquant-Surface and Bactiquant-Water). Each on-site training course lasts approximately 5 hours per course (“Surface” and “Air”) plus an additional 1-2 hours for porous materials. All courses end with a required final exam. We also offer a course on the Internet and on CD-ROM. This course covers all steps of sampling, analysis and test questions. In order to pass the final examination, at least 80% of the questions must be answered correctly. Our experience has shown that personal on-site training is the more effective option.
Mould infestation should always be treated, regardless of the type of mould. There may be species of fungi that have a greater impact on health than others. However, it is undisputed that there is no good mold if it occurs in too high a concentration indoors. Mold problems are primarily a question of quantity. The larger the regions affected by mold, the greater the problem. Dead mold should be removed just like viable mold. Mold remediation does not mean killing the mold, but (and this is more important) removing it.
The Equipment
For analyzing all types of samples
Waterproof case containing a handheld fluorometer, 3 rags, 2 pipettes, a thermometer, and a timer.
Proficiency training course (5 hours) included in the price. A small exam must be passed to obtain the proficiency certification number.